However, even with the additional polarity, this molecule will still remain neutral at physiological ph H. So we are able to label this amino acid as polar. So introducing it into the amino acid does introduce a molecular dipole.

And then we have our sulfur bonded to a hydrogen, sulfur is a relatively electro negative atom. Sistine introduces a sulfur in its structure in the long structure that comes off of a methyl group. Furthermore, the exposure, a bunch of hydrocarbons would make this amino acid relatively hydrophobic as well. So we can go ahead and label this as non polar. Benzene rings are only composed of carbons and hydrogen. So final valentine has a final group on it, which is basically just a benzene ring. Sorry, we're just going to better underline that. If you're trying to memorize these structures for the um cat. So I feel like that's a good way to remember it. So you can see where the name comes from, Final eleni. So we have our metal from our original group and then we have the final group added on there. Our next amino acid is final alunan phenylalanine introduces a final group attached to the alunan.
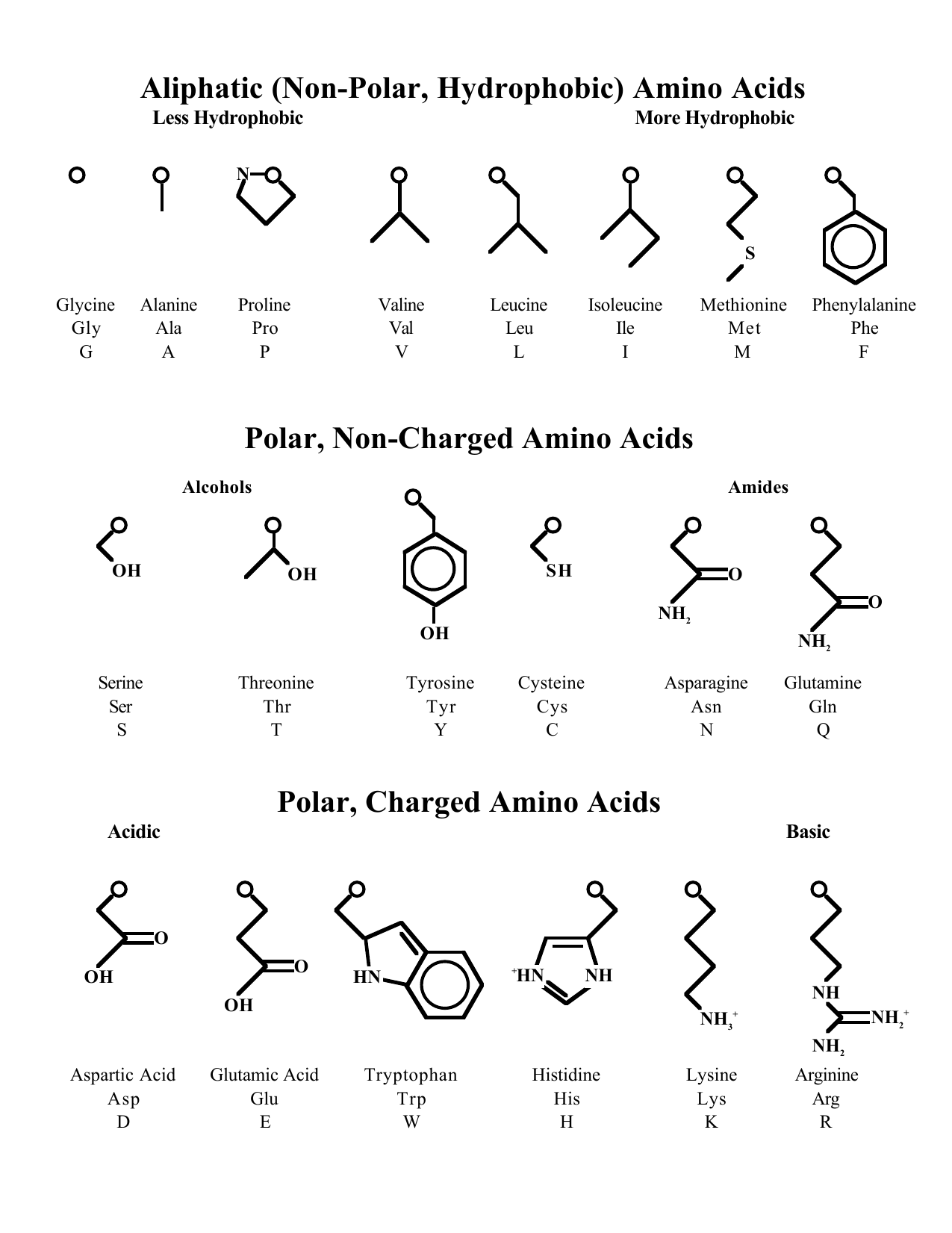
The our group similarly does not help the amino acid dissolve in water as it does not introduce an acidic or basic characteristic to the amino acids. So because it doesn't introduce any polarity into the molecule, we can label this as non polar based on its our group being non polar. This method group does not introduce any polarity into the molecule as it's relatively small and it's only an out pill group. So looking at um alan in the substitution on allen is a methyl group right here. So to start we have a line, we are supposed to first look at if the amino acid is polar, non polar hydrophobic or hydrophobic.

In this question, we're asked to look at three Amino acids and describe these amino acids and their characteristics.
